Books

Research Interests
Global biogeochemical cycles in the geologic
past and present;
geochemical and transport processes in the surficial and underground
environment; natural and anthropogenic controls of geochemical systems.
Research Projects
Biogeochemical
Cycles and Global Change
The
concept of the geochemical cycles of the
elements emerged in the middle to late 1800s, based on the studies of
the water
cycle from the 17th century,
later understanding of the chemical
composition of
waters, sediments, and crystalline rocks, and the studies of plant
photosynthesis since the late 18th
century. The modern
concepts of geochemical or biogeochemical
cycles date from the 1920s. We are studying the interactions
between the global biogeochemical cycles of the life-important
elements carbon, phosphorus, and nitrogen by means of conceptual
and mathematical models of these cycles that are compatible with the
available
data for the global C-N-P-O system in the remote and younger geologic
past. The industrial and agricultural byproducts of human activity
compete in their magnitudes with the sedimentary and geochemical
processes
on environmentally large scales. Human-made perturbations of the
geochemical cycles and their long-term consequences are being
studied at a global scale as well as within such specific environments
as the land and the oceanic coastal zone. The broader
goals
of this research, shown in somewhat more detail below, are to
understand the natural and human-induced global change in the past,
present, and future.
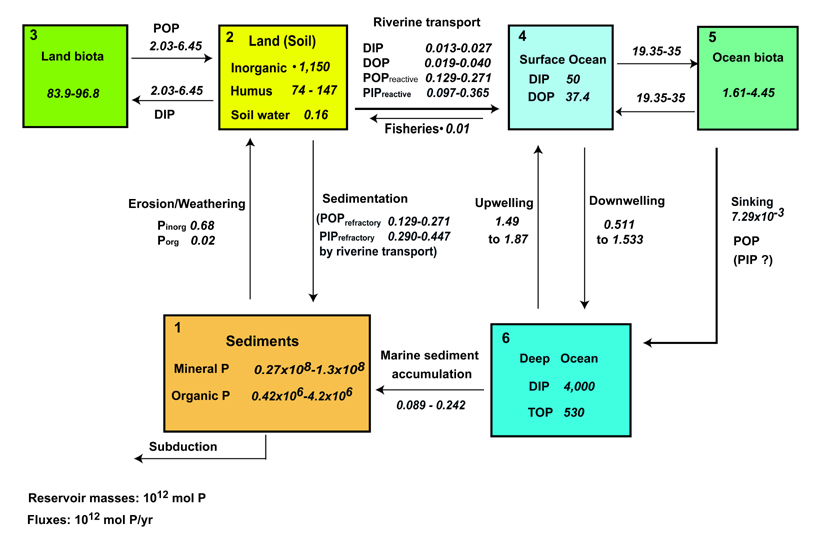
Global phosphorus
cycle at present
 Carbon
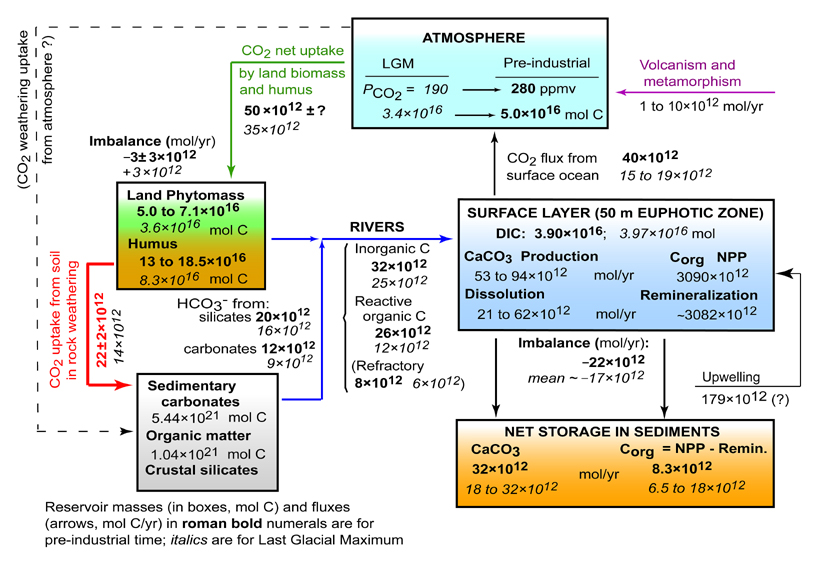
cycle since Last Glacial Maximum to Modern Time
Carbon
cycle since Last Glacial Maximum to Modern Time
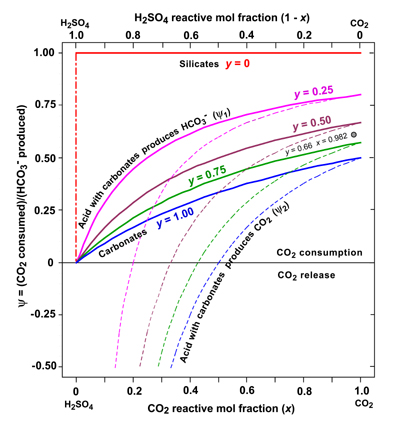
CO2
in weathering on land and river water composition
Carbonic
and sulfuric acids are the main inorganic acids that react with
minerals in the Earth's weathering crust. CO2
is produced by the oxidation of organic matter in the pore space of soils
and H2SO4
is the result of oxidation of mineral pyrite (FeS2).
The consumption of CO2 and production of HCO3‾
in weathering reactions depends on the carbonic/sulfuric acid ratio in
solution and the relative proportions of carbonate and silicate
minerals, expressed in the weathering potential, ψ,
that usually varies from 0.5 for pure carbonates to 1.0 for pure
silicates. This acid consumption model makes it possible to calculate the
chemical composition (metal cations and anions) of an average river
water, in very good agreement with other estimates.
Consumption of carbonic acid and sulfuric
acid, forming from pyrite, in the weathering of carbonates and silicates
|
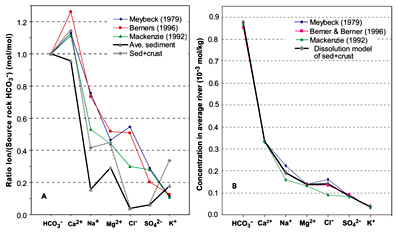
A.
World-average rivers (different sources), average sediment, and
sediment plus continental crust. B. Calculated ion concentrations
from CO2 and H2SO4 reactions with
sediments and crust.
|
|
|
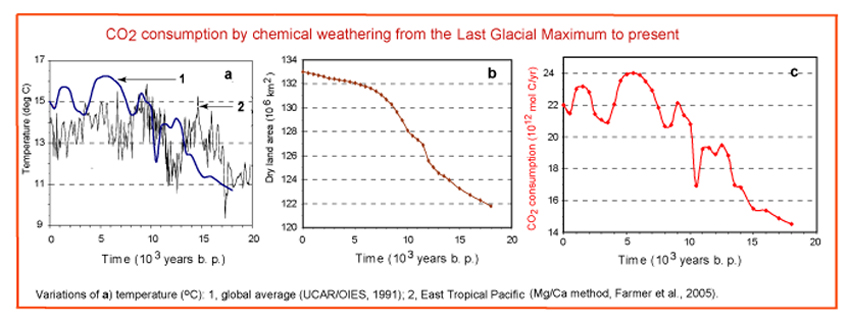
Since the Last Glacial Maximum, about
18,000 years ago, mean temperature of the Earth's surface has increased
by about 4°C and the dry land surface area increased about 10%. In
our estimation, these two factors were responsible for an increased
flux of CO2 to mineral weathering,
to its pre-industrial value of 22×1012
mol C/yr.
Human
perturbations of the global C-N-P cycles
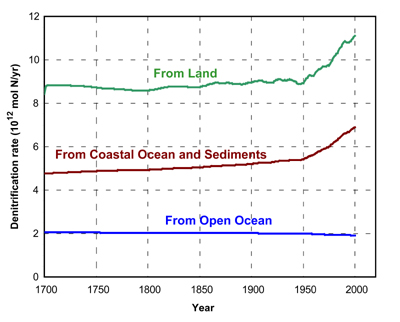
Interactions of the
global reservoirs of the atmosphere, land, coastal ocean, and open
ocean under the effects of the major human perturbations have resulted
in an increased transport of carbon, nitrogen, and phosphorus to the
coastal ocean, in particular since the mid-1900s. The analysis of the
global C-N-P cycles, using the models TOTEM (publications
1999-2005) and SOCM (publications
2004-2006) indicates increasing input and subsequent remineralization
of organic carbon in the coastal ocean, counteracting to some extent
the transport of CO2
from the atmosphere to coastal ocean waters. Among the results,
our estimates of the denitrification rates on land, in the coastal
ocean, and open ocean indicate a significant increase in industrial
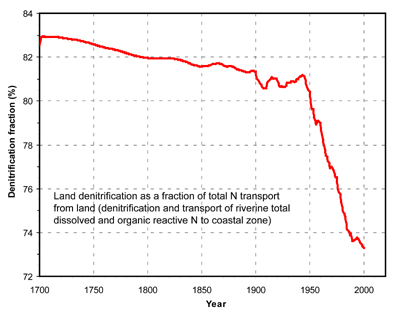
time. Because of the increasing erosion and decay of soil organic
matter, the release of N to the atmosphere by denitrification from land
became a smaller fraction of total N transport from land to the
atmosphere and coastal ocean.
40Ar/40K ratios as indicators of diagenesis
in sedimentary
sequences
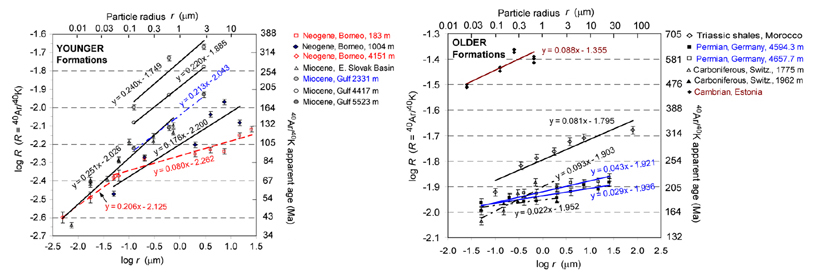
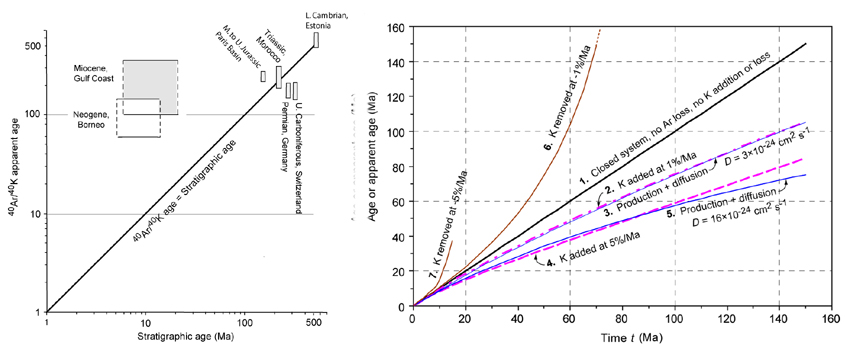
In potassium-bearing clay minerals,
the 40Ar/40K
ratio decreases with decreasing particle size, as has been documented
by a number of investigators. Smaller values of the 40Ar/40K ratio correspond to younger K/Ar ages or
apparent ages.
We
quantified the relationship between the apparent age (or the ratio R = 40Ar/40K) and the particle size in sediments ranging
in age from the Cambrian to the Neogene. A relationship of a type
log R = constant + b log r
makes it possible to determine the fraction of 40Ar
escaped from the smaller particles relative to the larger sizes. For
the sedimentary lithosphere in the Phanerozoic, we estimate the 40Ar
flux to the atmosphere at 6×106 to 17×106
mol 40Ar/yr, approximately 20 to
50% of the flux from the crystalline continental crust.
There are numerous occurrences of clays
that show K/Ar ages considerably older than the stratigraphic age of
their formations. This suggests that in many sedimentary environments,
clays have a long pre-depositional history and the decrease in the
apparent age and 40Ar/40K ratio in the fine-size clay particles are
due to the release of 40Ar.
However, a theoretical analysis shows that a decrease in the apparent
age may be due to addition of K to clays in the course of diagenesis, a
phenomenon that is difficult to detect. The fact that nearly the same
decrease in the apparent age may be achieved by addition of K, without
loss of 40Ar, as in a case of 40Ar production and simultaneous diffusion out
of particles, suggests that different diagenetic mechanisms might have
been active in different sedimentary sequences.
 Return
to Faculty &
Research
.
Return
to Faculty &
Research
.











